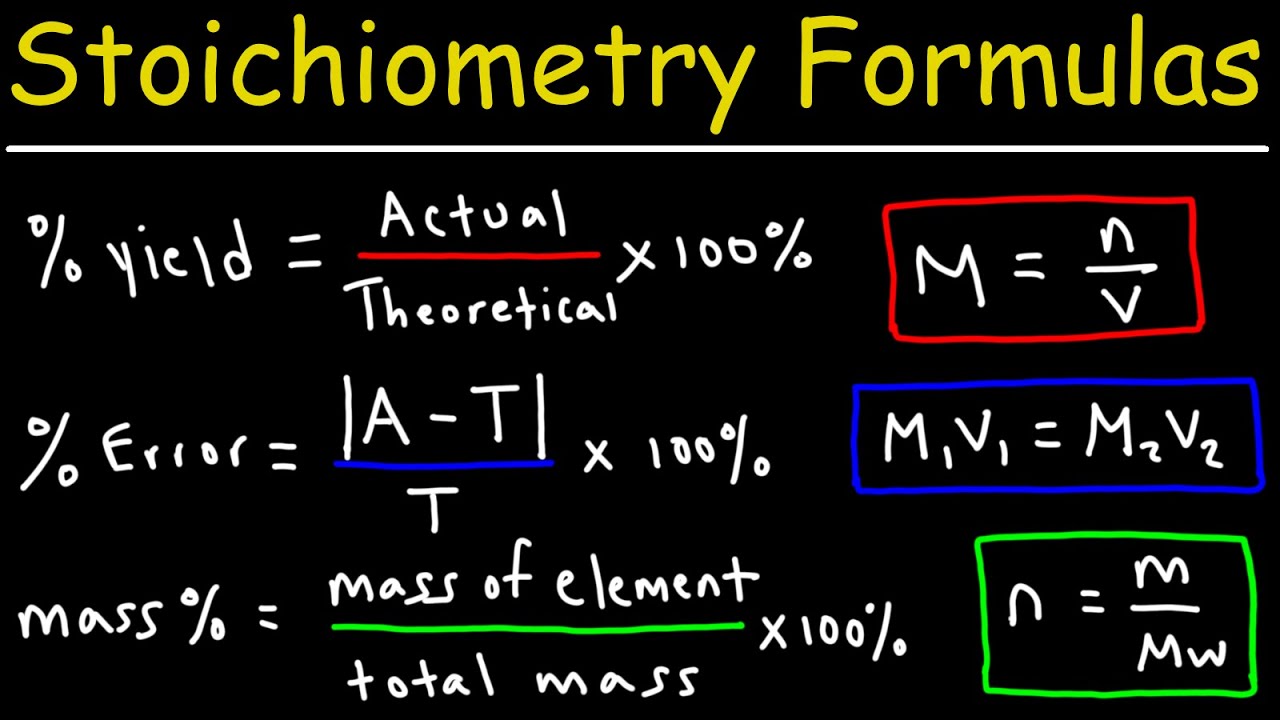
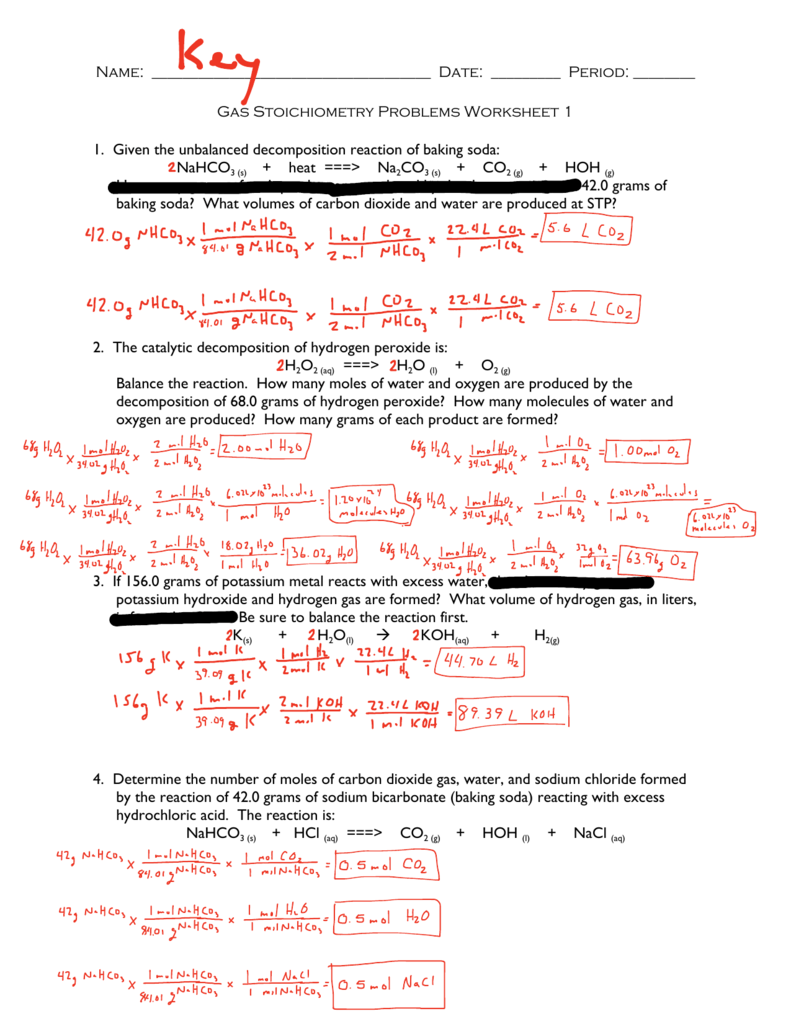Stoichiometry Sheet 6 Answers - From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? The decomposition of sodium azide, nan3(s), is used to inflate. 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10 and 45. Chapter 6 balancing and stoichiometry worksheet and key topics: • balancing equations • writing a chemical equation • stoichiometry practice: Base your answer to the following question on the information below. Stoichiometry with solutions name _____ 1.
Chapter 6 balancing and stoichiometry worksheet and key topics: 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10 and 45. From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? Base your answer to the following question on the information below. • balancing equations • writing a chemical equation • stoichiometry practice: Stoichiometry with solutions name _____ 1. The decomposition of sodium azide, nan3(s), is used to inflate.
2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10 and 45. Base your answer to the following question on the information below. From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? Chapter 6 balancing and stoichiometry worksheet and key topics: The decomposition of sodium azide, nan3(s), is used to inflate. Stoichiometry with solutions name _____ 1. • balancing equations • writing a chemical equation • stoichiometry practice:
Stoichiometry Practice Worksheets Answers Chemistry
Stoichiometry with solutions name _____ 1. Chapter 6 balancing and stoichiometry worksheet and key topics: • balancing equations • writing a chemical equation • stoichiometry practice: From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? The decomposition of sodium azide, nan3(s), is used to inflate.
Free Printable Stoichiometry Worksheets, 60 OFF
The decomposition of sodium azide, nan3(s), is used to inflate. 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10 and 45. Base your answer to the following question on the information below. Stoichiometry with solutions name _____ 1..
Worksheet For Basic Stoichiometry Printable And Enjoyable Learning
From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? • balancing equations • writing a chemical equation • stoichiometry practice: 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h.
Stoich w ssr_and_dr with key PDF Worksheets Library
2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10 and 45. Chapter 6 balancing and stoichiometry worksheet and key topics: Stoichiometry with solutions name _____ 1. Base your answer to the following question on the information below. From.
SOLUTION Chemistry stoichiometry practice problems with answers
Base your answer to the following question on the information below. The decomposition of sodium azide, nan3(s), is used to inflate. 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10 and 45. From the amount of nitric acid.
Stoichiometry Formulas and Equations College Chemistry YouTube
The decomposition of sodium azide, nan3(s), is used to inflate. From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10.
Stoichiometry Practice Worksheets Answers Chemistry
Chapter 6 balancing and stoichiometry worksheet and key topics: Base your answer to the following question on the information below. From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? Stoichiometry with solutions name _____ 1. 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h.
Stoichiometry Problems Worksheets
The decomposition of sodium azide, nan3(s), is used to inflate. From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? Stoichiometry with solutions name _____ 1. Base your answer to the following question on the information below. • balancing equations • writing a chemical equation • stoichiometry practice:
Stoichiometry Worksheets With Answers
Chapter 6 balancing and stoichiometry worksheet and key topics: • balancing equations • writing a chemical equation • stoichiometry practice: The decomposition of sodium azide, nan3(s), is used to inflate. 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h.
Mole Calculations 10th Grade Quiz Quizizz
Base your answer to the following question on the information below. The decomposition of sodium azide, nan3(s), is used to inflate. Stoichiometry with solutions name _____ 1. 2 c 6 h 10 + 17 o 2 12 co 2 + 10 h 2 o 8) if i do this reaction with 35 grams of c 6 h 10 and 45..
2 C 6 H 10 + 17 O 2 12 Co 2 + 10 H 2 O 8) If I Do This Reaction With 35 Grams Of C 6 H 10 And 45.
Stoichiometry with solutions name _____ 1. Base your answer to the following question on the information below. From the amount of nitric acid given in part a, how many moles of nitrogen monoxide will be made? The decomposition of sodium azide, nan3(s), is used to inflate.
• Balancing Equations • Writing A Chemical Equation • Stoichiometry Practice:
Chapter 6 balancing and stoichiometry worksheet and key topics: