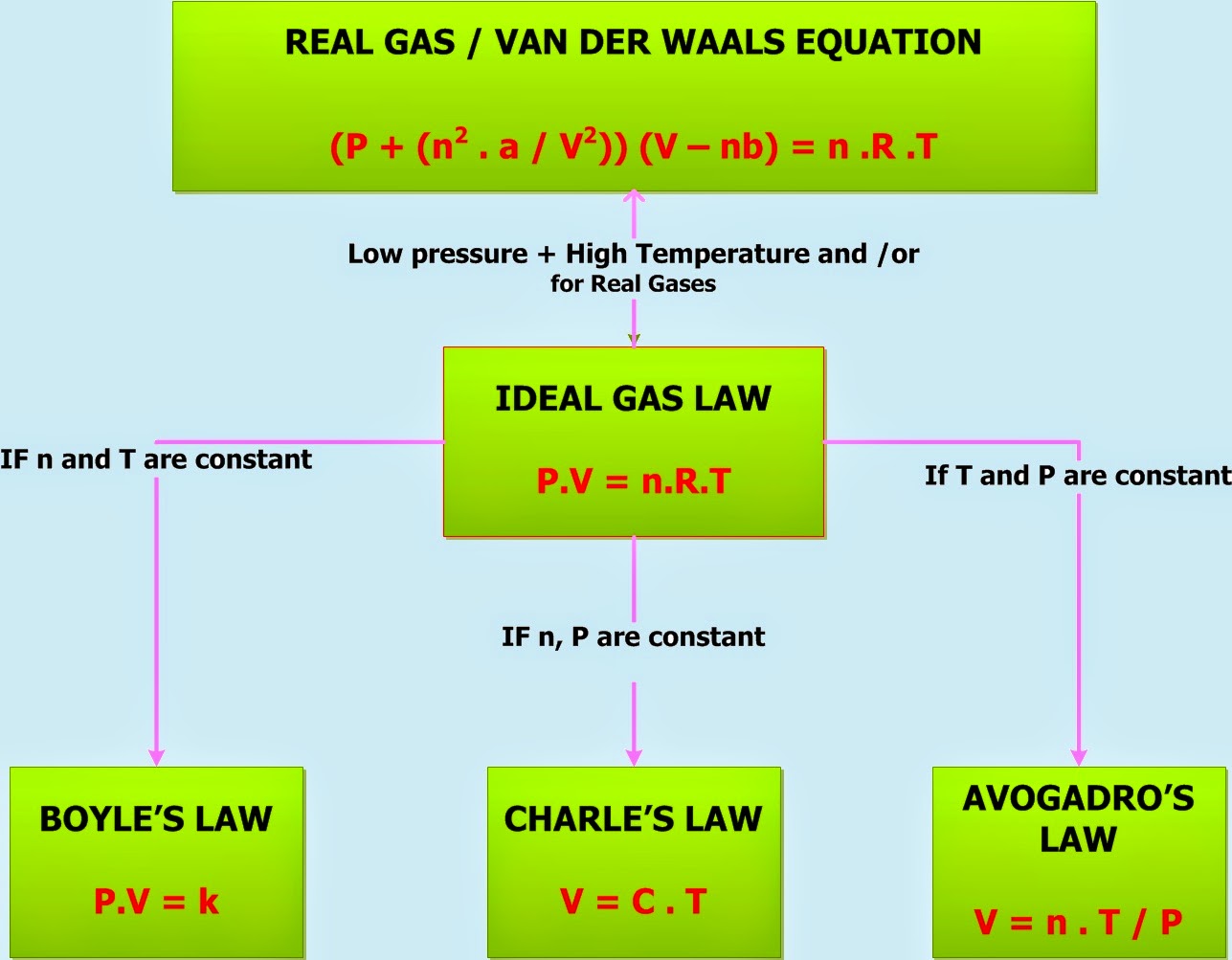
Gas Laws Formula Sheet - Gas particles are neither attracted to. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. Gases consist of large numbers of tiny particles, which have mass. The distance between particles is great. When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water.
When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to. Gas particles are neither attracted to. The distance between particles is great. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. Gases consist of large numbers of tiny particles, which have mass. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water.
Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. Gases consist of large numbers of tiny particles, which have mass. Gas particles are neither attracted to. The distance between particles is great. When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to.
Gas Law Formulas Sheet
When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. Gas particles.
Gas Laws Cheat Sheet Formulas & Constants
Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. The distance between particles is great. Gas particles are.
Gas Law Formulas Sheet
When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. The distance.
Chemistry GAS LAWS All Formulas for engineering& jee/neet 2021
Gas particles are neither attracted to. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. Gases consist of large numbers of tiny particles, which have mass. The distance between particles is great. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters.
How To Use Combined Gas Law at Joan Ray blog
Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. Gases consist of large numbers of tiny particles, which have mass. When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to. Ideal gas law pv = nrt.
Gas Law Formulas Sheet
Gas particles are neither attracted to. Gases consist of large numbers of tiny particles, which have mass. When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water..
All Gas Law Formulas
The distance between particles is great. Gas particles are neither attracted to. Gases consist of large numbers of tiny particles, which have mass. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters.
Gas Law Formulas Sheet
When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from the liquid’s vapor in order to. The distance between particles is great. Gas particles are neither attracted to. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. Ideal gas law pv =.
Gas Laws Equation Sheet Formulas & Conversions
Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. The distance between particles is great. Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. Gases consist of.
Ideal Gas Law — Overview & Calculations Expii
The distance between particles is great. Gas particles are neither attracted to. Gases consist of large numbers of tiny particles, which have mass. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. When collecting a gas over a liquid (usually water) you need to subtract how much pressure is coming from.
Gases Consist Of Large Numbers Of Tiny Particles, Which Have Mass.
Ideal gas law pv = nrt p = pressure in atm, kpa, or mmhg v = volume in liters n = number of moles t = temperature in kelvin ideal gas constant =. Gas particles are neither attracted to. Gas collection over water a mixture of gases results whenever a gas is collected by displacement of water. The distance between particles is great.